NOMIS Awardee Tony Wyss-Coray and colleagues have published research results that shed light on COVID-19 related neurological disease. The article, “Broad transcriptional dysregulation of brain and choroid plexus cell types with COVID-19,” was published in bioRxiv on Oct. 22, 2020.
Abstract
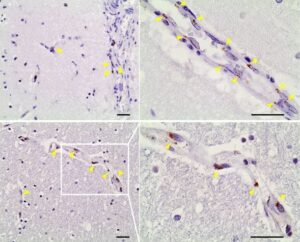
Though SARS-CoV-2 primarily targets the respiratory system, it is increasingly appreciated that patients may suffer neurological symptoms of varied severity1–3. However, an unbiased understanding of the molecular processes across brain cell types that could contribute to these symptoms in COVID-19 patients is still missing. Here, we profile 47,678 droplet-based single-nucleus transcriptomes from the frontal cortex and choroid plexus across 10 non-viral, 4 COVID-19, and 1 influenza patient. We complement transcriptomic data with immunohistochemical staining for the presence of SARS-CoV-2. We find that all major cortex parenchymal and choroid plexus cell types are affected transcriptionally with COVID-19. This arises, in part, from SARS-CoV-2 infection of the cortical brain vasculature, meninges, and choroid plexus, stimulating increased inflammatory signaling into the brain. In parallel, peripheral immune cells infiltrate the brain, microglia activate programs mediating the phagocytosis of live neurons, and astrocytes dysregulate genes involved in neurotransmitter homeostasis. Among neurons, layer 2/3 excitatory neurons—evolutionarily expanded in humans4—show a specific downregulation of genes encoding major SNARE and synaptic vesicle components, predicting compromised synaptic transmission. These perturbations are not observed in terminal influenza. Many COVID-19 gene expression changes are shared with those in chronic brain disorders and reside in genetic variants associated with cognitive function, schizophrenia, and depression. Our findings and public dataset provide a molecular framework and new opportunities to understand COVID-19 related neurological disease.
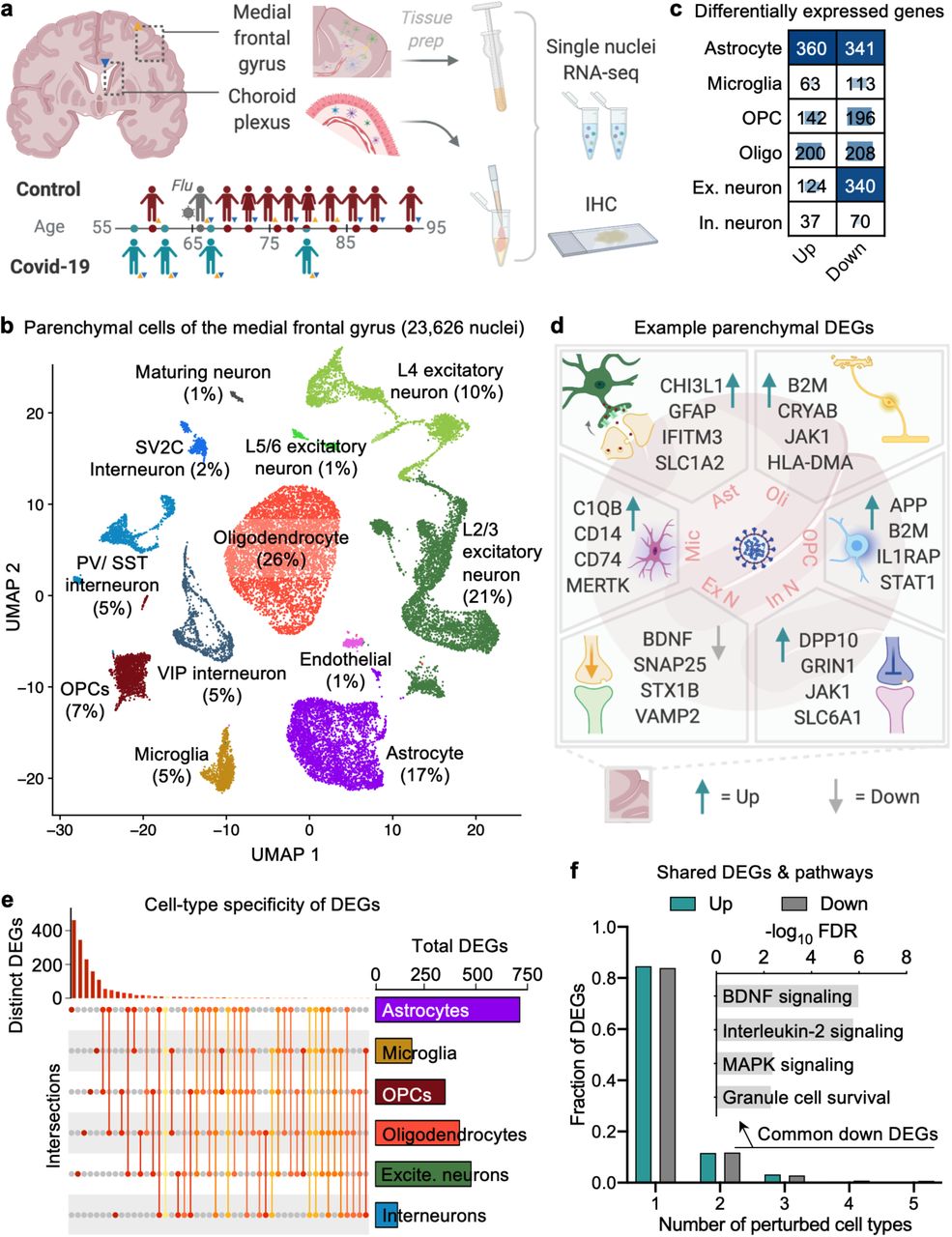
a, Study design. Colored triangles denote brain regions studied for each patient. b, Uniform Manifold Approximation and Projection (UMAP) of 23,626 nuclei captured from the human medial frontal cortex, colored by cell type and labeled with percent of total nuclei. Note, that as in prior reports32,34,35, the ‘Endothelial’ cluster also exhibits vascular mural cell markers. c, Differentially expressed gene (DEG) counts for each cell type (MAST with default thresholds, FDR < 0.05, Log (fold change) > 0.25 (absolute value)). The intensity of the blue color and the size of the squares are proportional to entry values. d, Example differentially expressed genes (DEGs) in COVID-19: excitatory (Ex N) and inhibitory (In N) neurons, astrocytes (Ast), oligodendrocytes (Oli), oligodendrocyte precursor cells (OPC), and microglia (Mic). e, Matrix layout for intersections of DEGs shared across and specific to each cell type. Circles in the matrix indicate sets that are part of the intersection, showing that most DEGs are cell type-specific. f, Fraction of total up- and downregulated genes (y-axis) as a function of the total number of cell types in which the differential expression occurs. Biological pathways associated with genes downregulated in COVID-19 that are common to ≥2 cell types are shown (n = 161 genes, hypergeometric test, FDR correction).
Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2), the etiologic agent of the COVID-19 global pandemic, has as of October 2020 infected over 40 million people and caused over a million deaths worldwide (worldometers.info/coronavirus). Though SARS-CoV-2 primarily targets the respiratory system, it is increasingly appreciated that COVID-19 patients can suffer neurological and psychiatric symptoms of varied severity, depending on SARS-CoV-2 mechanisms as well as other variables, such as co-morbidities and clinical care1–3,5. Central nervous system (CNS) symptoms range from loss of smell and headache to memory loss, encephalitis, stroke, difficulty concentrating, and persistent fatigue1,6–8. Symptoms are especially frequent and prominent in the over 20% of COVID-19 patients that require hospitalization1,9–11. With lingering CNS symptoms documented in Severe Acute Respiratory Syndrome (SARS) and Middle East Respiratory Syndrome (MERS) survivors1,12–14, it remains to be seen whether such chronic symptoms will emerge in COVID-19 survivors.



